Purpose
The purpose of this lab is to determine the ratios of moles to reactants in a chemical equation when the formulas of the products are not known. In this lab, sodium and hypochlorite and sodium sulfate were to be reacted together and the temperature tested. By mixing different ratios of each chemical, the goal is to find the best-fit point where the temperature produced by the reaction will be the highest, utilizing the most of the reactants. Using this information, the optimum ratio, or stoichiometric ratio will be found, creating the balanced chemical equation.
Data Table
|
|
|
|
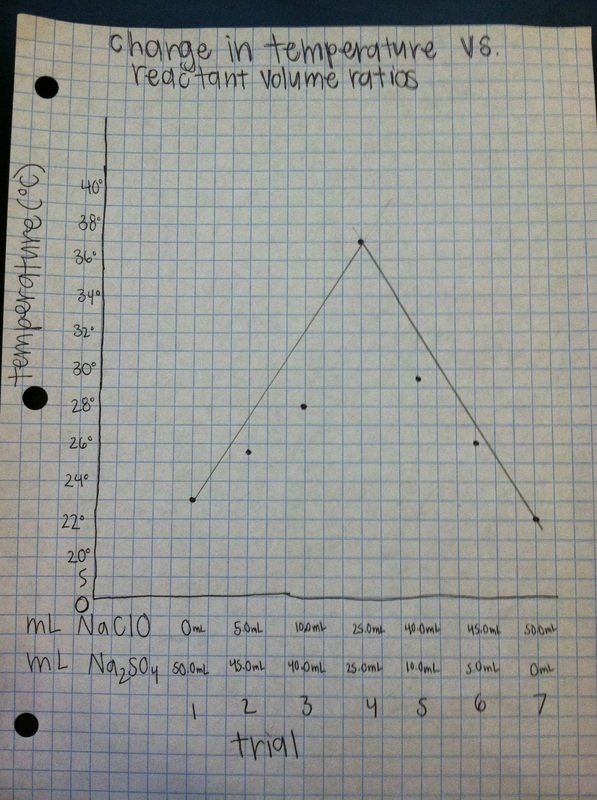
Graph
Conclusion
In conclusion, a balanced chemical equation gives the mole ratios of reactants and products for chemical reactions. If not all of the formulas are known, experimental measurements must be made to determine these ratios. In this experiment, the stoichiometric ratio for the reactants sodium hypochlorite and sodium sulfite are unknown. When different ratios of the two substances were combined in a reaction, the temperature was measured, and the optimum ratio of the two substances that was determined was 25.0 mL of both NaClO and Na2SO3 will provide the highest temperature, 37°C. With this ratio, the maximum amount of reactant will be utilized during the reaction, which causes the higher temperature to be produced.
Discussion of Theory
By combining different ratios of the two given chemicals in this reaction, a fairly precise stoichiometric ratio is found. The answer is evident when the temperatures produced by each reaction are plotted on a graph. The higher the point is on the graph, meaning the higher the temperature produced, the more reactants being used between the two substances, sodium hypochlorite and sodium sulfite. All of the reactions occurring are exothermic, therefore the heat produced will be directly proportional to the amount of reaction that occurs. During a reaction, the maximum amount of reactants that can be used will be used, and the chemical equation is left with the optimum ratios of moles used. This experiment is a sort of guess-and-check method of finding this stoichiometric ratio. As long as the total volume is kept constant at 50mL, any combination of the two reactants can be tested to find the best two volumes that work best together. This method is called the method of continuous variations. When the optimum ratio is acquired, the greatest amount of reactants is consumed, forming the largest amount of product possible given the starting volumes of the reactants. It all works out in the end because not only is there the best ratio found from proof in the reaction, but the lines of best fit on the graph provides the same answer, so it is known that the correct answer was arrived at.
Analysis Questions
- The reactants had to stay at a constant total volume because if they weren't the amount of heat produced would not directly correlate with the amount of the reactants that reacted, it wouldn't be even and line up.
- A limiting reagent is the compound in an equation that is completely used up. It prevents the reaction from reacting even more because it gets used up the fastest.
- Volume limits precision because it is the independent variable. The person choosing the ratios of each reactant to use in the experiment is limited in finding the exact answer. The temperature is the dependent variable, so whatever ratio is chosen to be reacted, a precise answer will come out based on what was put in.
- Along the upward slope of my graph, the limiting reagent is sodium hypochlorite. Along the downward slope, it is sodium sulfite.
- The method of continuous variations could also be used in reactions involving change in color intensity, the mass of a precipitate formed, or the volume of a gas created.
- It is more accurate to use the point of intersection because it represents the the theoretical point where the optimum ratio would exist. The ratio associated with the greatest temperature change may not be accurate because a similar ratio may have not been tested, but could produce a greater temperature change than one that was tested during the experiment.