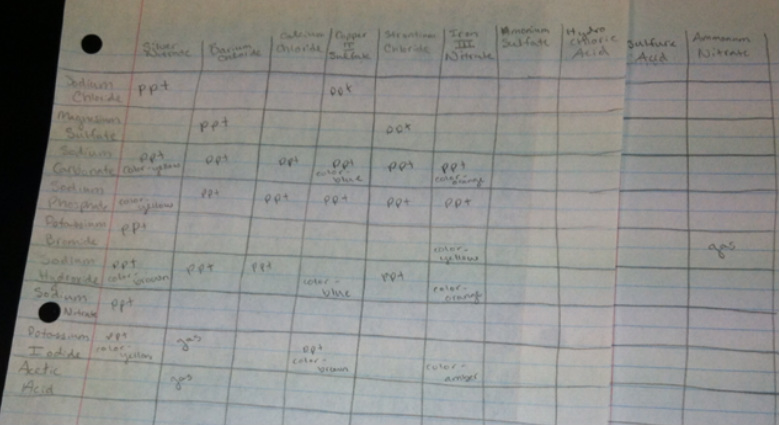
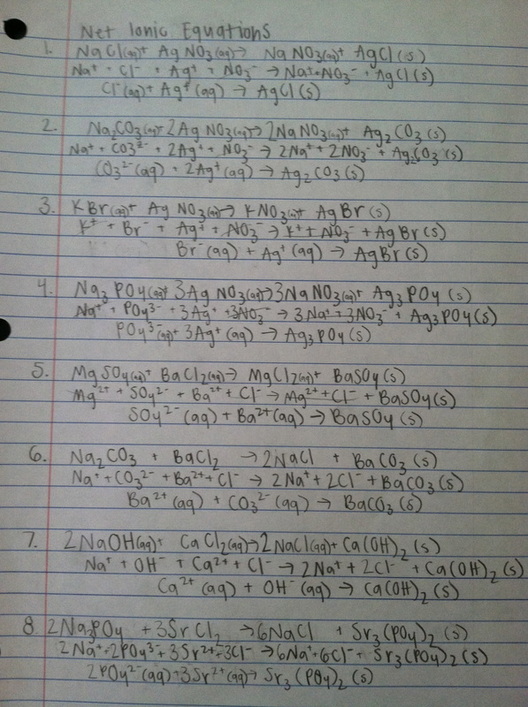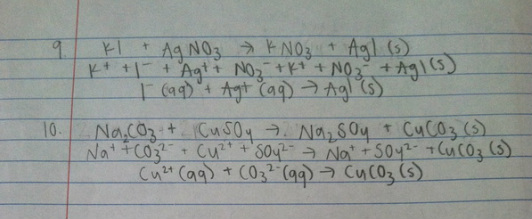Data Table
Conclusion
Throughout the the ninety possible reactions that were tested between the mixing of 19 chemicals, 31 reactions appeared to have taken place. Between different chemicals, certain trends seem to have occurred throughout the reactions. Almost all of the reactions involving silver nitrate took place, making it the most reactive chemical in the group, forming precipitates, many of them color changing. This was able to happen because silver is an exception for many of the solubility rules, creating insoluble products. The chlorides were semi-reactive and created mostly precipitates. The transition metals resulted in numerous color-changing precipitate reactions. All nitrates besides silver were fairly non-reactive because they all create soluble products. In addition, all acid and ammonium based reactants did not react at all except for the creation of a gas on two different occasions. Lastly, when a color change took place, the color that appeared as a result of the reaction stayed fairly constant through all of the reactions using the column of a chemical on the top row on the data table.
Discussion of Theory
When ninety different chemical reactions take place at once, it is evident that they will all have different varying outcomes. Many of them will not react at all. This is because the solubility rules won't allow it. A double replacement reaction such as the ones in this lab won't take place when both of the products are soluble substances. This happens because in water, all of the ions are floating around separately, and no new compounds are able to form this way. However, when one of the products of the reaction is an insoluble compound, a precipitate, or solid substance will form within the aqueous solution, resulting in the reaction taking place and completing. The polarity of each molecule affects whether or not it will react. The higher the polarity, the more soluble the substance is, and the less likely it is to be able to react in the equation. That being said, the lower the polarity, the less soluble the substance is and the more likely it is to react and form a precipitate because the ions will combine in the solution and form a new compound. Besides forming a precipitate in the reaction, there is a possibility for a gas to be formed or a neutralization to occur when the reaction completes. A gas can be formed from the two reactants during a reaction, however it is the most uncommon type of reaction to occur. A neutralization reaction happens when water and an ionic salt are produced as a result of a double replacement reaction such as the ninety that took place in this lab. Each compound used in a set of reactions will react differently, but will usually follow trends. For example, one compound might be very reactive because of its ability to form insoluble substances, causing all reactions using that compound to occur and form a precipitate. If one of these compounds tends to cause a color change throughout the course of the reaction, the color will usually be pretty similar for each reaction that occurs with it. While this uniformity is shown, it may also be shown by a compound's tendency to not react at all because of its solubility or otherwise. The net ionic equations of each reaction taking place out of a large group of reactions in an experiment will help to clarify the specifics of the outcome of each mixture of two compounds, and show why what happens, happens.
Sources of Error
There are many things that could've caused inaccurate results in this experiment. If there was air in the dropper when the chemicals were dropped onto the transparency, bubbles would appear in the pool of chemicals, making it appear to have reacted forming a gas, when it really didn't. Also, since there were so many reactions to take place, and all of them were so close together and the chemicals are all clear, one may have been dropped in the wrong square on the chart, which would leave us with inaccurate results of what reacted and what didn't. In addition, for this same reason, the recorder may have misrecorded the results of one of the reactions because the boxes are in such close proximity that it would be easy to write something in the wrong box. The test size of each reaction, being so small, may not have been enough to see a visible result if the color, precipitate, or gas produced wasn't very prominent or large. This would have caused the group to not see a reaction that really did occur.
Analysis Questions
1. According to my data acquired from this lab, it appears that the cation silver is generally insoluble, the cation barium is generally insoluble, the cation calcium can go either way, the cation copper is generally insoluble, the cation strontium can go either way but more times than not it is insoluble. The cations ammonium and sulfate were very soluble, and so was the cation sodium for the most part. The anion hydroxide was generally insoluble, and the anion acetate was soluble. Anions carbonate and phosphate were both very insoluble. Bromides and chlorides were both generally soluble as well.
2.
2.
3. The first type of metathesis reaction is a precipitation reaction. In these, a solid, non-soluble product is formed. It occurs when two soluble ionic compounds are mixed together, they exchange partners and one of the products is an insoluble compound so it drops out of solution as a solid. An example of a precipitation reaction from this lab is (NaCl + AgNO3 --> NaNO3 + AgCl). The next type of metathesis reaction is the formation of a weak or non-electrolyte, when the formation of water and a salt occur through neutralization. An example of this type of reaction from the lab is
(HCl + NaOH --> H2O + NaCl). The final type of metathesis reaction is the formation of a gas. This is the least common of the metathesis reactions. It occurs when one of the metathesis products is a gas with a low solubility in water. Generally one of the reactants will be an acid. An example of a reaction from this lab where a gas was formed was (KBr + H2SO4 --> SO2 + KHSO4)
4. The solubility of a substance is determined by its polarity. The more groups of polar molecules that a compound contains, the more soluble it is in water. These groups can form hydrogen bonds with water, which are very strong. Because these bonds are very strong a molecule that contains more polar groups such as OH- and NH2- is more water-soluble. In addition, solubility is determined by a maximum amount of solute that dissolves in a solvent at equilibrium, or a state where reactants and products reach a balance - no more solute can be dissolved in the solvent in the set conditions, creating a saturated solution where nothing more can dissolve in the water.
5. A double replacement reaction that produces two aqueous products is considered a "no reaction" because nothing is formed from the reaction. When the reactants are mixed together in the solute, they aren't really compounds. It is really the individual ions just floating around like a soup. The ions can be mixed around and rearranged but they can't join together and form a precipitate or new compounds from the reactants in the equation.
(HCl + NaOH --> H2O + NaCl). The final type of metathesis reaction is the formation of a gas. This is the least common of the metathesis reactions. It occurs when one of the metathesis products is a gas with a low solubility in water. Generally one of the reactants will be an acid. An example of a reaction from this lab where a gas was formed was (KBr + H2SO4 --> SO2 + KHSO4)
4. The solubility of a substance is determined by its polarity. The more groups of polar molecules that a compound contains, the more soluble it is in water. These groups can form hydrogen bonds with water, which are very strong. Because these bonds are very strong a molecule that contains more polar groups such as OH- and NH2- is more water-soluble. In addition, solubility is determined by a maximum amount of solute that dissolves in a solvent at equilibrium, or a state where reactants and products reach a balance - no more solute can be dissolved in the solvent in the set conditions, creating a saturated solution where nothing more can dissolve in the water.
5. A double replacement reaction that produces two aqueous products is considered a "no reaction" because nothing is formed from the reaction. When the reactants are mixed together in the solute, they aren't really compounds. It is really the individual ions just floating around like a soup. The ions can be mixed around and rearranged but they can't join together and form a precipitate or new compounds from the reactants in the equation.