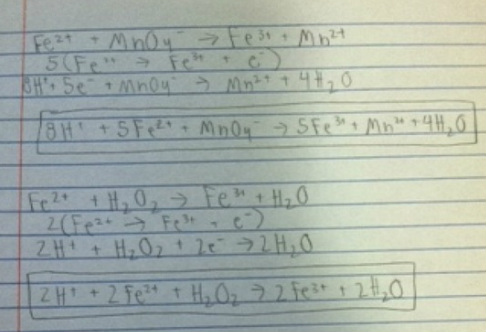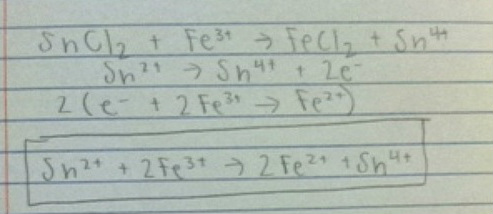Purpose
Three experiments will be run where a compound which is colorless in solution when reduced is converted to a deeply colored solution when oxidized. The complete balanced reactions for each step should be written showing the transfer of electrons during oxidation and reduction. This lab will demonstrate the transfer of elections in an oxidation-reduction reaction as one element gains and one element loses them.
Data Table
|
Part of Lab
Part 1 Part 2 |
Observations
-When the 10mL of the potassium thiocyanate soltuion was added to each beaker, they both turned peach in color -When potassium permanganate was added to the beaker labeled KMnO4 and mixed, it instantly turned a redish brown black color and the dark color remained -When hydrogen peroxide was added to the beaker labeled H2O2 and mixed, it instantly turned a dark auburn color and stayed dark -When stannous chloride solution was added to each beaker, the KMnO4 beaker stayed a dark brownish color, but it got a little lighter, and the H2O2 beaker got a little lighter, and the color became redder and brownish -When the potassium hydroxide solution was diluted with de-ionoized water with 5 g of dextrose dissolved into the solution and serveral drop sof methylene blue solution added, the solution turned a dark blue color -After the flask stood for 5-10 minutes untouched, ths osolution got a lot lighter, and almost clear |
Conclusion
At the start of the first reaction, the two beakers containing the ferrous ammonium sulfate were clear in color. When the potassium thiocyanate was added to each, the color turned a clear peachish tint. When the potassium permanganate was added to its respective beaker, the color instantly turned into a reddish brown black color that stayed dark. When the hydrogen peroxide was added to its respective beaker, it also instantaneously turned into a color that appeared blackish red, somewhat like blood. After the stannous chloride was added to each beaker and mixed, the one with the KMnO4 stayed dark brownish, but it got a little lighter in color than it was before. The beaker with the H2O2 began to get lighter and redder and browner in color. In a separate reaction when potassium hydroxide solution is diluted with de-ionized water, it appeared clear in color. when dextrose was dissolved into it, the color remained clear. However, after several drops of methylene blue were added to the solution, it turned dark blue in color. After the flask stood untouched for about 10 minutes, the solution got lighter and almost clear in color. After the flask was vigorously shaken, though, it became blue again. The change from clear to dark shades in each of the solutions in these reactions occurred because there was oxidation and reduction taking place.
Discussion of Theory
A redox reaction is an equation that contains the oxidation of one element and the reduction of another element. Oxidation is the gain of oxygen, and reduction is the loss of oxygen. When oxygen reacts with another element in a reaction, it will gain electrons (except for fluorine), as it is taking the electrons away from the element that it is reacting with. However, with this being said, oxygen is not always a component in redox reactions. With this in mind, a better way to describe oxidation is as a loss of electrons, meaning the charge increases, and reduction as the gain of electrons, where the charge decreases. These two things always happen together, as one can not occur without the other. Redox reactions are similar to acid-base reactions in that respect, because they are always found in pairs. In addition, electrons are never lost or gained during a redox reaction. Any electrons that are lost by a certain molecule are always picked up by another, hence the oxidation and reduction always going together. This is also where the name "redox" comes from, as a mixture between "oxidation" and "reduction" combined together in the name like they always are in the reaction. When redox reactions are created, they begin as two half reactions combined into a whole. One half reaction is an oxidation reaction and the other half is a reduction reaction. One half loses electrons and the other gains them, respectively. These types of reactions are typically very useful in batteries and power cells, for example galvanic cells that have one size containing the negative anode where oxidation occurs and the other size containing the positive cathode where reduction occurs. These two halves are connected by a salt bridge and the complete oxidation-reduction reaction is complete and can run throughout. To ensure that the same number of electrons are being lost and gained by each half reaction, the number of electrons being exchanged is always balanced out. Algebraically, the electrons on each side will cancel each other out and leave the final net ionic equations with no greater or fewer electrons. In many cases, the effects of a redox reaction can be seen physically. Because of this, it can be much more simple to reverse the reaction that occurred, because the results are physically measurable. In this lab and the three redox reactions that occurred, the effects of the oxidation and reduction were clearly visible in the change in color that happened. When the color changed from nonexistent, or clear, to dark auburn or blue depending on which reaction was taking place, it was evident that the oxidation and reduction had taken place. In the first two reactions, the solution turned a red color because oxidation of Fe2+ containing compound to Fe3+ in the presence of thiocyanate will become red in color. In the methylene blue solution, it turned blue because the oxidation of the dye methylene blue keeps a deep blue color, because methylene blue is a leuco dye, which is a dye whose molecules can exists in two forms, one form colorless (seen when the solution sat still) and one form colored (seen when the solution was shaken).
Data Analysis Questions
1.
At room temperature, the standard reduction potential of the permanganate reaction is 1.51 volts, and for the hydrogen peroxide equation, the potential is 1.77 volts. This means that the hydrogen peroxide has a higher reduction potential and the reaction it is involved in is more likely to occur and result in the H2O2 being reduced than the permanganate reaction that occurred.
2.
2.
(Spectator ions were excluded from net ionic equations in numbers 1 and 2)
4. When the colorless solution of methylene blue is shaken, it becomes dark blue in color. This is because the oxygen in the air oxidizes the solution when it is shaken to make the color react in the solution and become visible. After the solution rests for an adequate amount of time, the methylene blue is able to react with the leftover amount of dextrose in the solution to become clear, and the process is repeated.
5. After 10-12 cycles, the methylene blue solution no longer returns to the blue color after resting after being shaken. This occurs eventually because every time that the solution is shaken and oxidized, it loses electrons. After the electrons are taken away so many times, it eventually runs out and cannot return to the blue color that it was previously.
4. When the colorless solution of methylene blue is shaken, it becomes dark blue in color. This is because the oxygen in the air oxidizes the solution when it is shaken to make the color react in the solution and become visible. After the solution rests for an adequate amount of time, the methylene blue is able to react with the leftover amount of dextrose in the solution to become clear, and the process is repeated.
5. After 10-12 cycles, the methylene blue solution no longer returns to the blue color after resting after being shaken. This occurs eventually because every time that the solution is shaken and oxidized, it loses electrons. After the electrons are taken away so many times, it eventually runs out and cannot return to the blue color that it was previously.